( C) CAR engineered T cells have a chimeric cell receptor with an scFv composed of variable heavy and light chains (V H and V L) of an antibody as the extracellular portion fused to intracellular T-cell signaling domains to cause T-cell activation upon interaction with the targeted cell surface marker. ( B) TCR engineered T cells have the addition of a second set of TCR α and TCR β genes (highlighted in red and pink) which are present in addition to the naturally occurring TCR α and TCR β genes (highlighted in blue). Genome editing is also being explored to enhance the survival and/or efficacy or prevent self-targeting of both natural (circulating T cells and TILs) and engineered (TCR and CAR) T cells. ( A) Genome editing (highlighted by green arrows) is being explored to create universal donor T cells to serve as the basis for TCR and CAR T-cell engineering. In this review, we examine the current status and scientific basis of clinical trials featuring ZFNs, TALENs, and CRISPR-based genome editing, the known limitations of CRISPR use in humans, and the rapidly developing CRISPR engineering space that should lay the groundwork for further translation to clinical application.ĬRISPR clinical trial gene activation genome editing transcription activator-like effector nucleases zinc finger nuclease. Current research addressing these issues may provide new opportunities for use of CRISPR in the clinical space. 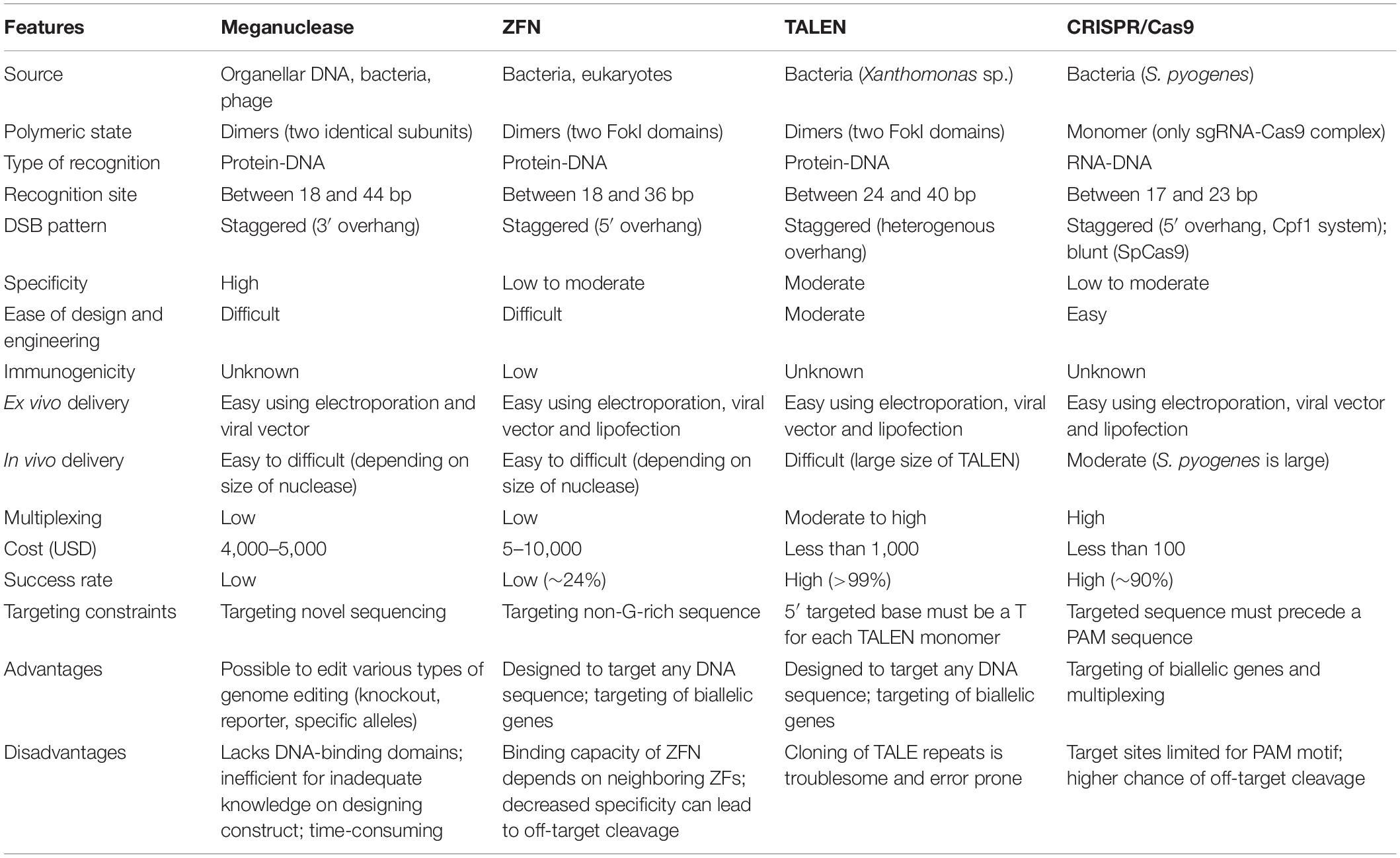
However, in vivo use of CRISPR technologies can be confounded by problems such as off-target editing, inefficient or off-target delivery, and stimulation of counterproductive immune responses. The ex vivo editing approach is highly effective for many disease states, including cancers and sickle cell disease, but ideally genome editing would also be applied to diseases which require cell modification in vivo. Most clinical use of CRISPR to date has focused on ex vivo gene editing of cells followed by their re-introduction back into the patient. Genome editing technologies, particularly those based on zinc-finger nucleases (ZFNs), transcription activator-like effector nucleases (TALENs), and CRISPR (clustered regularly interspaced short palindromic repeat DNA sequences)/Cas9 are rapidly progressing into clinical trials.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
